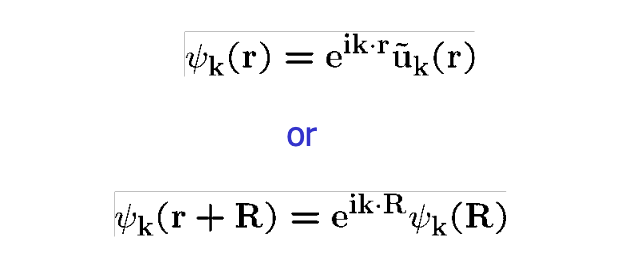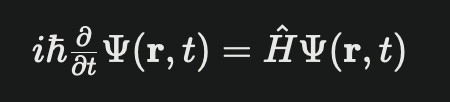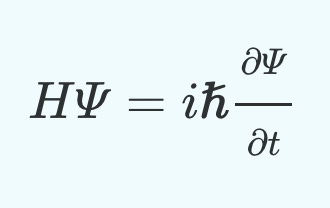bloch's theorem
I’ve recently encountered Bloch's theorem while reading solid state physics. While I’ve taken a detour from solid state physics to think about some more machine learning based ideas, I wanted to circle back now to breakdown what this equation is, and why it’s interesting. Formally:
Bloch's theorem states that solutions to the Schrödinger equation in a periodic potential can be expressed as plane waves modulated by periodic functions.1
Or stated mathematically:
If this sounds and looks like gibberish to you, you’re in the right place! Let’s break down everything here, and hopefully by the end of this you’ll understand what’s going on.
Schrodinger’s Equation
Let’s start by defining Schrodinger’s equation. The Schrodinger’s equation defines the evolution over time of the wave function. Mathematically, the equation is the evolution over time of the wave function.
Let’s break down everything here.
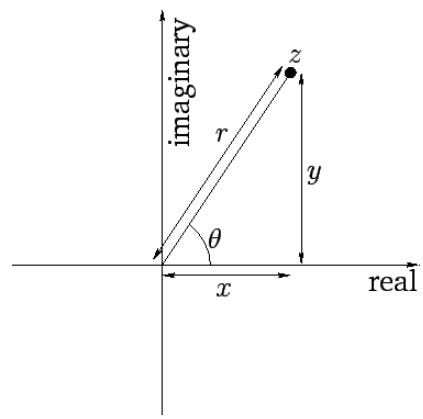
i is the imaginary unit. It may seem odd to have something called “imaginary” describing real physical phenomenan, but “imaginary” numbers are just as real as real numbers! It is a handy algebraic object to define a vector of size two, a real and imaginary component. Put another way, we are attempting to describe one number with two dimensions.
Here we can see how the equation z = r * cos(θ) + i * r sin(θ) enables us to encode two dimensions, the real and the imaginary, into one algebraic expression with one variable r by changing the coordinate system. For waves, we use it to describe two different properties in one function: amplitude (real part A) and frequency - phase (imaginary part kx-wt).
ψ(x,t) = A[cos(kx-ωt) + i sin(kx-ωt)]or with euler’s theorem
ψ(x,t) = Ae^i(kx-ωt)We will revisit this representation method more when we talk about the wave function. For now, the i serves to ensure that our end equation will have an imaginary part, and thus properly represents a wave. There are other necessary reasons why, such as the unitarity, but they all derive from the fact we are trying to represent a wave.
ℏ is Planck’s constant. Planck’s constant is a number dervied by empircal observation. It’s just an axiom of our universe that pops up when we measure elementary particles. Here it pops up because the energy of a system is directly proportional to the wavelength times the Planck constant, which is exactly what Schrodinger’s equation is getting at. This, again, is just a nature of our universe that these two values, energy and wave frequency, are related in this way.
∂/∂t, just means a partial derivate, or a derivative with multiple variables. Here it is because we have two variable time and space. The derivative itself is the chage in the wave fuction, whose inputs are time and space, with respect to time. Another equivalent writing of the schrodinger equation that makes this more clear is:
Ψ(r,t) is the wave equation. We can think of the wave function as the probability distribution or particles in space and time. Mathematically, remember from our discussion above on i, we generally use complex numbers to represent waves. Here our equation is no different than any other wave we represent in this way.
Ψ(r,t) = Ae^i(k·x - ωt)Where A is the amplitude (real portion), while k and w are the frequency and phase (imaginary portion) with respect to space and time (r, t).
Ĥ is the Hamiltonian operator. The hamiltonian operator is just another term for the total energy of a system. Here we have both kinetic energy, because the particles are moving, and potential energy, because the particle is usually in a field like gravity or a magnet. Each has their own complex mathematical representation, but for brevity just think of the hamiltonian operator is all the energy in a system. When we multiply this by the wave function, we get the expected value of the system's total energy.
We now have a breakdown of every component in the equation! Here it is again:
We can now see why we define this equation as such. That the change in the wave function with respect to space and time (the left side) is equal to the expected value of the system's total energy applied the existing wave (the right side)!
If this is not making sense, consider the mechanical equivalent. If I have someone on a swing, and push them, the change in their position and velocity is contigent on the energy I have put into the swing. Here we are saying the same thing, that the change in the wave fuction, which is just the probability distribution or particles in space and time, is contigent on what energy is being applied to the system.
So whe we say we are “solving” for Schrodinger equationn, we are essentially calculating all the energy in the system, and then seeing how the probability distribution of an elementary particle, in this case electrons, moving through it changes as a result of adding energy. When we look at solid state physics, we see that this starting energy comes from the density of ions in the solid which form a periodic potential.
Periodic Potential
Solids are dense! Meaning they have lots of ions in a very small amount of space. This density creates uniformity, usually in the form of a bravais lattice. This uniformity + density thus means that the energy each ion exerts, creates a uniform set of potential energy fields. When electrons travel, they travel through these fields. This creates the basis of energy of our schrodinger’s equation (the right side) that the electrons add to.
When we say we are solving the Schrodinger’s equation in a periodic potential, we are asking that as the energy of our system changes from adding electrons (the right side), how does the distribution of those electrons moving through the solid change (the left side)? As it turns out, it does so in plane waves.
Plane Waves in Periodic Functions
The objective of any physics theory is to create a mathematical model that explains empircal findings. Why should electrons be able to travel through some solids well, and some solids poorly? Well Schrodinger’s equation says that the energy of a system should relate to the wave function. So if the material is creating some kind of energy potential, it must have an effect on the wave fuction!
So what is a plane wave? Well a plane wave is just a wave, like the equation we saw when discussing the wave function in Schrodinger’s equation. When we discuss the plane wave, we are describing the energy and position of electrons within a system. These two ideas, energy and position, are intimately related, but that’s a blog post for another time. The electrons on their own would have some kind of wave equation to describe their energy, how would this change as a result of being in a solid? Thakfully, the answer is relatively clear, it’s jus the wave equation times whatever the periodic potential is.
This makes sense! Electrons traveling through a solid should spread out throughout that solid, so when we think about what the energy of the electrons should be, it should reflect the available space within the solid that it is passing through! We can now turn to the actual Bloch equation:
Which says exactly what we have stated before. The wave function, aka the thing on the right side of the Schrodinger’s equation is equal to a plane wave, which is just a representation of the electrons momentum & energy represented using imaginary numbers, times the existing potential, which is just how the added existing energy due to the layout of the solid.
These periodic function are also handy because they provide us an algebraic form of the basis for the wave form of electrons in a solid. A basis of a solid is just how the ions in the particle are spread out. Since the periodic function represents the wave form of those ions, it makes sense that it is a good representative of the basis of a solid.
With this equation, we can now see why some solids are good conductors, while others are not. The ability for particles to have momentum and direction inside of a solid, is directly related to how the ions are laid out in the solid. If the ions are too packed together, the periodic fuction will dampen the wave function of the electrons, thus not readily allowing electorns through. If the periodic functionn does have nice consisten gaps, with beneficial potentials, then electrons can more easily move through. This is also why chips and fragmented solids are bad conductors, or can ruin the conductiveness of a solid. The chip messes with the periodic structure of the material, and thus can alter the periodic function of a solid which has a direct effect on the waveform of electrons.
Conclusion
I think the point of this entire article is less about Bloch’s theorem, and more example of
How complicated quantum theory makes everything
You aren’t stupid, you just don’t have context
Even simple looking theorems stand on a mountain of science. Just compiling all that information to build enough understanding for one function is incredible. Having that entire context is difficult, but once achieved allows you to understand vastly more complicated ideas.
https://en.wikipedia.org/wiki/Bloch%27s_theorem