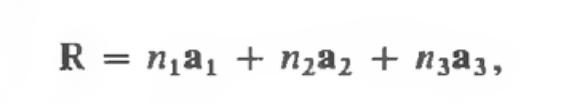bravais lattice
I’ve been reading solid state physics, and been struggling mightily with it. To my credit it’s a dense book filled with niche concepts that haven’t been relevant since the 70s. In any case, I wanted to take a few posts to confirm my understanding of some of the material. The book seems to care about two important attributes of matter. The first are properties given external stimulus such as heat and electricity. The second is the actual structure of atoms in the material. Most notably is the Bravais Lattice, a crucial idea in crystal physics. I’m going to take the article to discuss what the Bravais Lattice is.
What are solids?
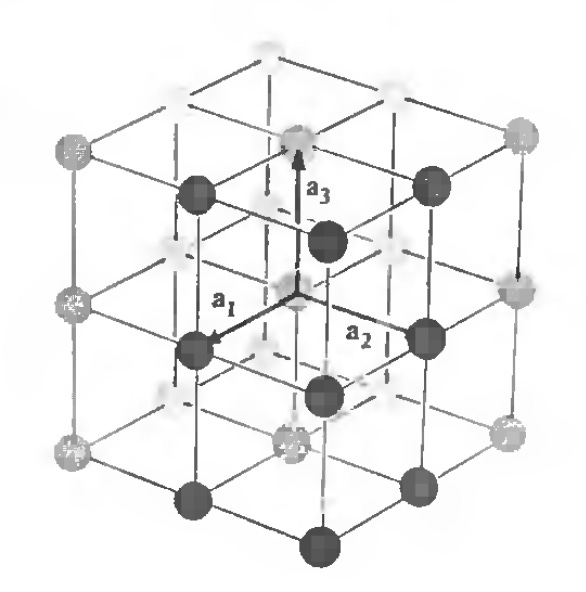
It may surprise many that most metals found in nature are crystals. While this crystal form gets refined away, the raw metal begins as a crystal. Their structure is one of an infinite array that appears is exactly the same no matter how it’s observed. We can see a 3d example here:
We see that each point is spaced exactly the same. While real solids are finite, this mathematical tool assumes that it’s an infinite space for ease of calculation. We can define the set of points as translations from a point R using vectors. We think of the equation as:
Where R is the resulting point after applying operations in the x, y, and z direction of the lattice. Taking the full set of combinations from this function, we see how these primitive vectors (a1, a2, a3) generate the lattice. Picking a random point R, we can select any combination of the vectors above function (so a1, a2 + a3, etc), and we must arrive at another point in the lattice.
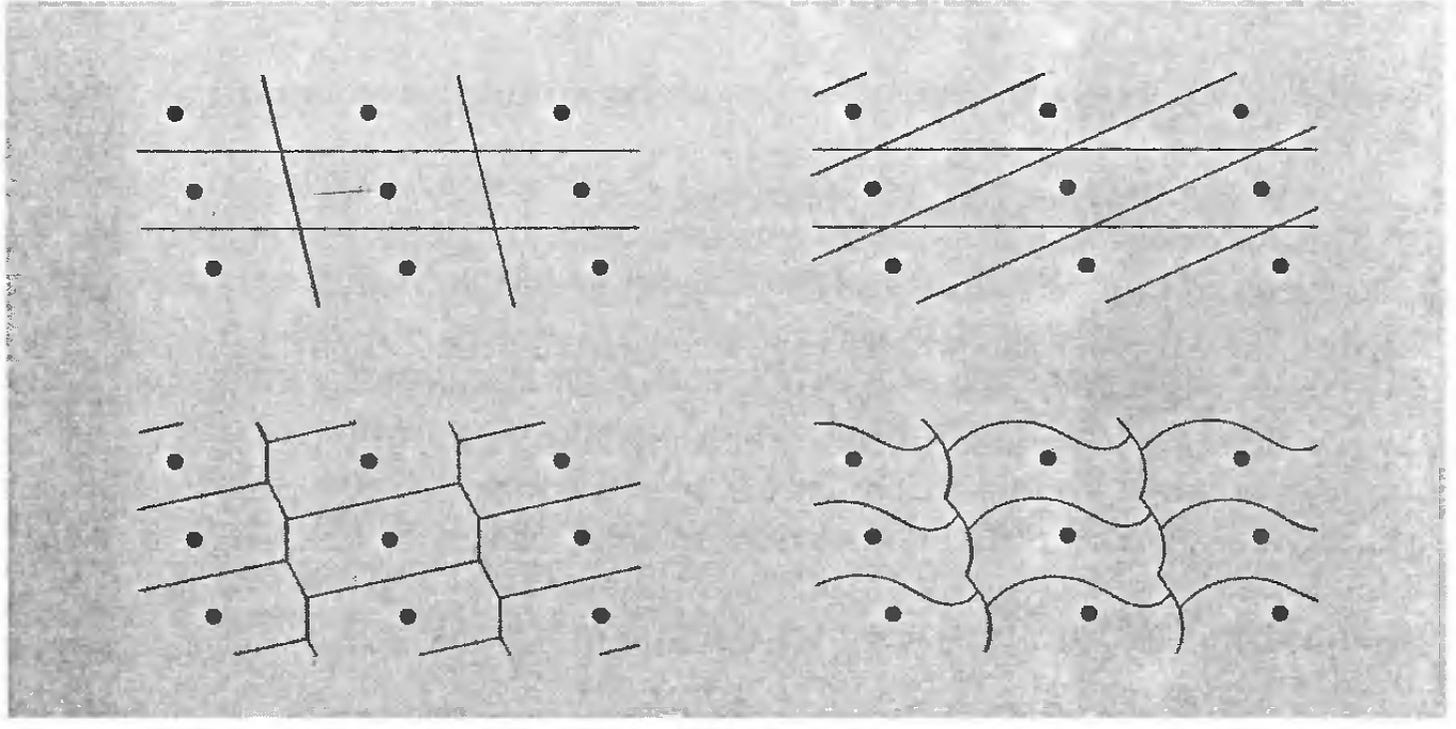
But a crystal structure isn’t just it’s atoms, it’s also the space between atoms. We call the minimum volume of space that each contain a single atom to be a primitive unit cell, or unit cell for short. There can be lots of possible unit cells:
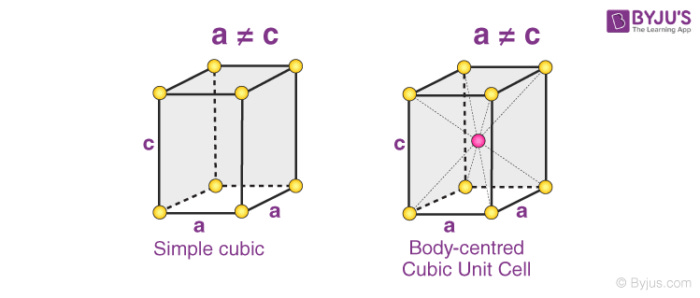
How do we select a unit cell? Well usually the nature of the crystal chooses for us. For example, some crystals display a square based crystal structure
while others might display more of a rectangular shapes
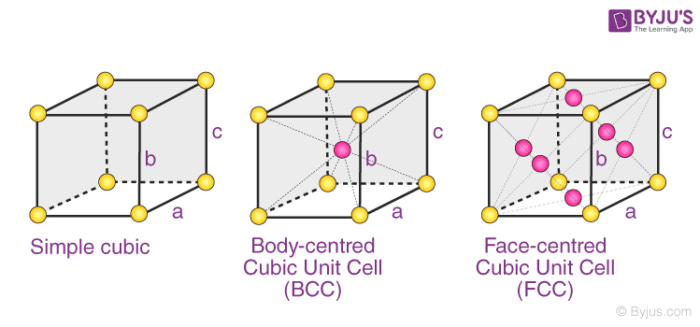
Notice too that we can have body centered or simple version s of the same unit cell. In simple we contain no atom. In body centered we contain one atom. In face centered, we contain one atom, and we have an atom on ever single face. Notice here again, each one of these keeps consistent with vectors.
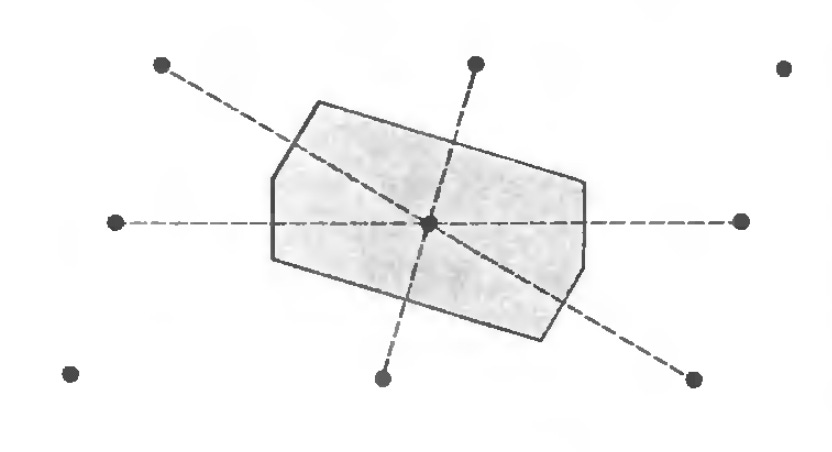
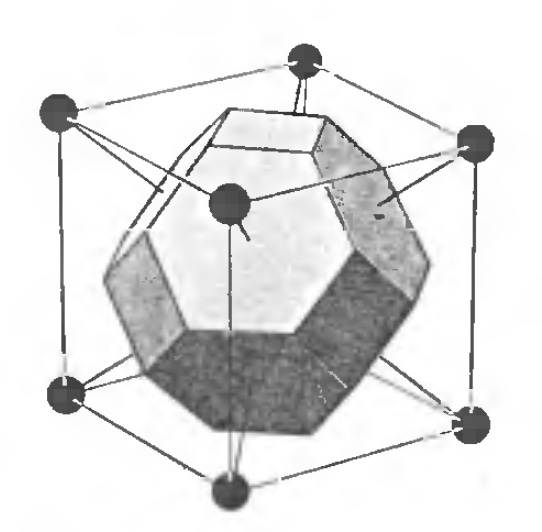
Another common strategy is the Wigner-Seitz Primitive Cell, which is to just calculate the nearest neighbors distance between each point. This results in a shape of size n, where n is the number of nearest neighbors. Here we see two examples of Wigner-Seitz primitive cells in 2 and 3 dimensional cells
The last important point is that these latices are based upon real atoms! While some will only contain one, such as pure iron, others may contain a combination. We call the combination of elements, ions, and other molecular players the basis of the latice. A basis plus it’s lattice is the crystal structure of all solids!
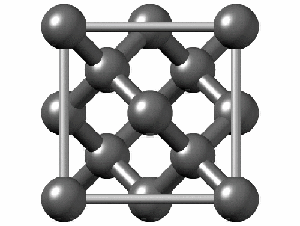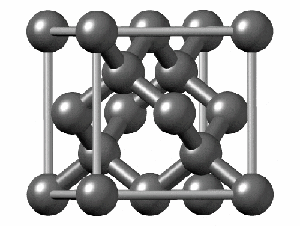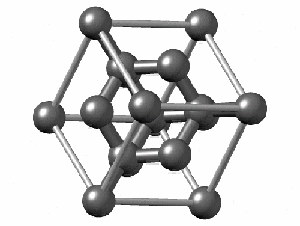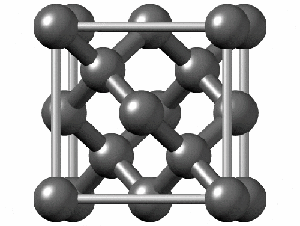
Not all elements form a Bravais Lattice. For example, Diamonds techincally don’t form a Bravais Lattice because their structure is too complex for a simple math object. They are formed by two Bravais Lattices that intertwine. Here we can see how complex the structure is:
Conclusion
While a lot of posts on tiling are strictly mathematical, the Bravais Lattice has a clear purpose. To describe the structure of a solids. It’s wild to me that nature has so much clear structure, and gets me thinking about how the refinement processes we use reshape this structure into whatever we need!